- Home
- Online Doctor
- Weight Loss
- Mounjaro Weight Loss Medication
FAQs
How Mounjaro® Works
Mounjaro is a once-weekly dual GIP/GLP-1 receptor agonist. It dials down hunger, slows gastric emptying, and helps regulate blood sugar — so you feel fuller, eat less, and lose weight alongside a reduced-calorie diet and movement.
Proven Benefits
Serious weight loss: Avg −15% to −21% at 72 weeks in adults without diabetes (dose-dependent). New England Journal of MedicinePubMed
Beats semaglutide 2.4 mg head-to-head at 72 weeks (−20.2% vs −13.7%). New England Journal of Medicine+1PubMed
Metabolic wins: Lower waist, BP, lipids; improved glycaemic control.Dosing & Administration
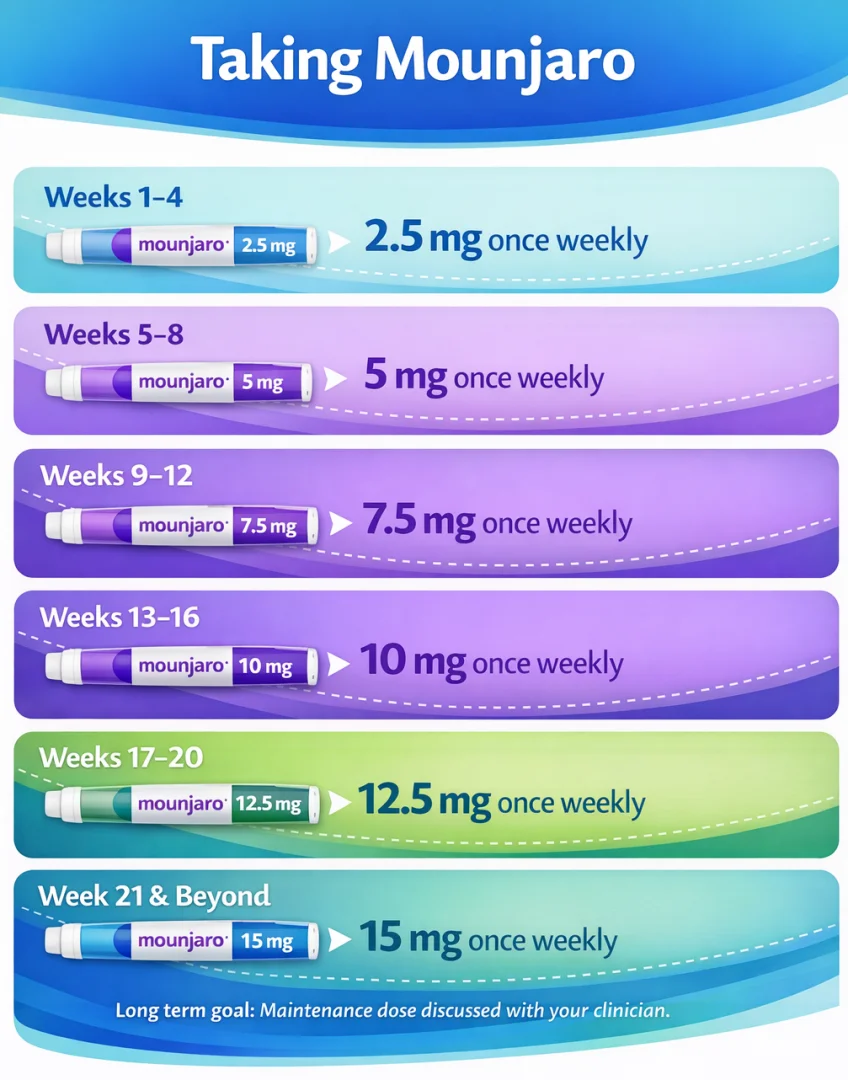
Start 2.5 mg once weekly for 4 weeks, then increase every 4 weeks (5 → 7.5 → 10 → 12.5 → 15 mg) as tolerated. Inject subcutaneously into abdomen, thigh, or upper arm. If you miss a dose, take it within 4 days; otherwise skip and continue your usual day.
Who Should Not Use Mounjaro
Under 18s (not studied)
Pregnant, trying, or breastfeeding — not recommended; use effective contraception (oral methods can be less reliable during initiation and dose increases).
Allergy to tirzepatide or ingredients.
Always tell your prescriber about all medicines you take.Possible Side Effects
Common: nausea, vomiting, diarrhoea/constipation, tummy pain (usually ease as doses step up).
Serious but uncommon/rare: pancreatitis, gallbladder issues, severe allergic reaction — seek urgent help if severe abdominal pain or signs of anaphylaxis. Report side effects via MHRA Yellow Card. PubMedLilly UKExpected Results & Timelines
Appetite drops in weeks 1–4; weight change typically shows in the first month, with steady, clinically significant lossbuilding across months as doses increase. Trial data show sustained results through 72 weeks (and maintenance with ongoing therapy).
Dosing Schedule
Weeks 1–4: 2.5 mg once weekly
Weeks 5–8: 5 mg once weekly
Weeks 9–12: 7.5 mg once weekly
Weeks 13–16: 10 mg once weekly
Weeks 17–20: 12.5 mg once weekly
Week 21 onwards: 15 mg once weekly (max; your prescriber may keep you at a lower maintenance dose) Lilly UK
Your Recently Viewed Items